Common menu bar links
Breadcrumb Trail
ARCHIVED - Assisted Human Reproduction Canada - Report
 This page has been archived.
This page has been archived.
Archived Content
Information identified as archived on the Web is for reference, research or recordkeeping purposes. It has not been altered or updated after the date of archiving. Web pages that are archived on the Web are not subject to the Government of Canada Web Standards. As per the Communications Policy of the Government of Canada, you can request alternate formats on the "Contact Us" page.
Minister’s Message

It is my pleasure to present the 2012-13 Report on Plans and Priorities for Assisted Human Reproduction Canada (AHRC), the federal regulatory agency established to protect and promote the health, safety, dignity and rights of Canadians who use or are born of assisted human reproduction (AHR) technologies.
AHRC continues to undertake its responsibilities under those provisions of the Assisted Human Reproduction Act that are in force and provides meaningful information to those Canadians and professionals impacted by the Act. It does so through a judicious use of its funds in ensuring that its activities are carried out in a cost effective manner and in support of government priorities.
The Government of Canada will take steps to meet its continuing responsibilities under the Act while respecting the opinion of the Supreme Court of Canada on the constitutionality of certain provisions of the Act. I am confident that AHRC will help advance the health, safety, dignity and rights of Canadians building their families through AHR and of those born from such technologies.
The Honourable Leona Aglukkaq, P.C., M.P.
Minister of Health
Section I: Organizational Overview
Raison d’être and Responsibilities
Assisted Human Reproduction Canada (AHRC) was created in response to a 1993 recommendation from the Royal Commission on New Reproductive Technologies, which called for the Government of Canada to establish a national agency to provide a safe and ethical framework for assisted human reproduction (AHR) activities and related research. Legally established under the Assisted Human Reproduction Act in 2006 and opening its doors in 2007, AHRC is mandated to administer and enforce the AHR Act and related regulations in order to protect and promote the health, safety, dignity and rights of Canadians who use or are born of assisted reproductive technologies.
In 2012-13, as the government considers the SCC opinion, AHRC will continue in accordance with the Assisted Human Reproduction Act to:
- Promote compliance and enforce the AHR Act related to the prohibitions;
- Provide information to the public and to the professions respecting assisted human reproduction and related matters, and respecting risk factors associated with infertility;
- Monitor and evaluate developments within Canada and internationally in assisted human reproduction and related matters;
- Consult persons and organizations within Canada and internationally; and,
- Provide advice to the Minister on assisted human reproduction and related matters.
These activities contribute to the Government of Canada's whole-of-government framework commitment to "Healthy Canadians" by helping to maintain and improve Canadians' health.
Strategic Outcome(s) and Program Activity Architecture
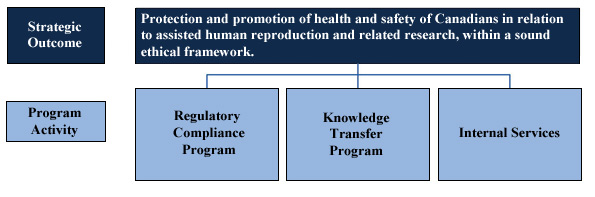
Organizational Priorities
Priority - Promote and enforce the legislation and regulations
Type1: Ongoing
Strategic Outcome(s) and/or Program Activity(ies): Regulatory Compliance Program
Status
Why is this a priority?
Promoting compliance and enforcement of the Assisted Human Reproduction Act, a legal instrument and legislation that underpins the raison d'être and responsibilities of the Agency.
Plans for meeting the priority
- Continuing to develop comprehensive tools to inform and assist relevant parties such as lawyers, medical practitioners, embryologists, patient groups, and the public in understanding and complying with various aspects of the AHR Act.
- Continuing identification, communication and compliance monitoring activities with regards to the prohibitions.
- Continue to collaborate with law enforcement authorities and professional bodies to educate them about, and engage them in enforcing, as necessary, the prohibitions of the AHR Act.
Priority - Foster the application of ethical principles in all facets of AHR activity
Type2: Ongoing
Strategic Outcome(s) and/or Program Activity(ies): Regulatory Compliance Program
Status
Why is this a priority?
Canadians want assurances that the technological innovations pertaining to AHR are consistent with their values. AHRC seeks to educate through the use of comprehensive tools these innovations in order to build a higher level of understanding amongst a diverse stakeholder community. AHRC must achieve a balance which upholds Canadians' ethical standards and which promotes the rights and dignity of AHR users and offspring.
Plans for meeting the priority
- Ongoing work to establish mechanisms and networks that provide guidance and facilitate best practices in order to protect and promote the rights and dignity of those impacted by AHR.
- Continuing to integrate ethical considerations, aligned with Canadian values, into all its relevant activities to reflect the sensitive nature of AHR and to uphold the rights and dignity of AHR users and offspring.
Priority - AHRC is a centre of expertise on AHR
Type3: Previously Committed to
Strategic Outcome(s) and/or Program Activity(ies): Knowledge Transfer Program
Status
Why is this a priority?
Transfer of knowledge pertaining to AHR is critical for medical stakeholders, patient groups and the general Canadian public.
Plans for meeting the priority
- Continuing to develop strategic relationships domestically and internationally to exchange AHR-related information/knowledge in order to facilitate national consensus and best practices that are evidence-based to safeguard health and safety while protecting values and ethics.
- Ongoing work to serve as a single, centralized, integrated entity keeping abreast of research and innovations in AHR, assessing evidence regarding health and safety risks, and disseminating knowledge to professionals, patient groups and the general public.
- Continuing to expand and enhance education and outreach activities accessible to all Canadians in order to raise awareness of infertility risks and inform of potential risks associated with AHR technologies.
Priority - Achieve Excellence in Governance and Management
Type4: Ongoing
Strategic Outcome(s) and/or Program Activity(ies): Internal Services
Status
Why is this a priority?
AHRC is committed to the judicious use of its resources and ensures that its activities are carried out in a cost-effective manner. As prescribed by the AHR Act, the Board of Directors is responsible for the overall management of AHRC including providing advice to the Minister of Health, approval of AHRC's goals, operational policies and budget, and the evaluation of AHRC's performance.
Plans for meeting the priority
- Continue to engage the Board of Directors in developing its planning and reporting instruments including strategic planning, and integrated business and human resources planning.
- As the Agency evolves, staffing continues to be aligned based on its role and responsibilities. AHRC's Internal Financial Governance and Control framework is used to maintain prudent financial management in accordance with Treasury Board policies.
Risk Analysis
reproduction (AHR), there remain health and safety risks to Canadians who use or are born of AHR technologies.
A recent study investigating the prevalence of infertility in Canada shows that an estimated 16%, or one in six couples5, suffers from infertility (defined as a failure to conceive after trying for a period of 12 months). There has also been a significant increase in the use of AHR in Canada, which has important implications for individual and public health, including an increased number of preterm or multiple births. AHRC has responded with a focus on knowledge transfer and stakeholder partnerships in areas such as: facilitating the development and promotion of a Canadian Framework with national targets to reduce multiple births that are a result of AHR; developing a web-based multiple birth patient portal to educate and inform Canadians; producing information products about infertility, using donated gametes or donating gametes to a third party and creating fact sheets to promote a better understanding of the prohibitions under the AHR Act.
Rapid technological advances may present health, safety, ethical and social challenges, and illustrate the importance of integrating ethical considerations into AHR activities. AHRC has kept pace with such developments: continually scanning the horizon to identify the latest scientific developments and their implications; developing credible, evidence-based health information; and liaising with other jurisdictions engaged in these issues, domestically and internationally.
Growing numbers are travelling to foreign destinations to access AHR services, which may present both health risks for patients and their offspring and health costs for Canadian society. AHRC is working with professional associations to raise awareness of risks and to promote evidence-based, informed decisions by Canadians considering cross-border reproductive care.
The Supreme Court of Canada opinion declared parts of the AHR Act unconstitutional, thereby significantly curtailing many activities that had been envisioned. At the same time, those aspects of health protection and promotion related to AHR that remain a federal responsibility are complex issues, often with ethical, legal, compliance and social dimensions. At the same time, other parties may be called upon to address Canadians expectations for oversight in the field of AHR that fall outside of federal jurisdiction.
Planning Summary
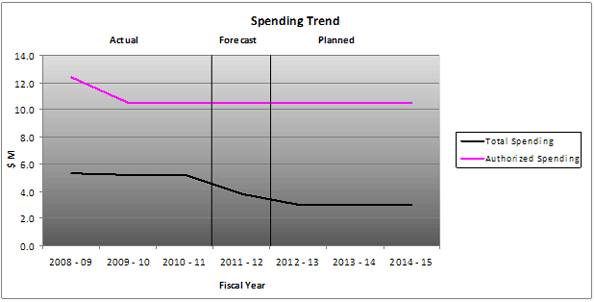
Assisted Human Reproduction Canada is allocated a resource envelope of $10.5 million for fiscal year 2012-13. Of this amount, $9.9 million requires approval by Parliament and the remaining $614,600 represents a statutory forecast of employer related expenditures that do not require additional parliamentary approval and is provided for information purposes. Since its inception, AHRC's expenditures have been well below its annual parliamentary allocation of $10.5 million.
The following table summarizes AHRC's total authorized spending for the next three fiscal years. While AHRC is authorizedto spend $10.5 million and staff 44 FTEs, the Agency anticipates its expenditures will be less than $3 million and staff less than 12 FTEs for 2012-13.
| 2012-13 | 2013-14 | 2014-15 |
|---|---|---|
| 10.5 | 10.5 | 10.5 |
| 2012-13 | 2013-14 | 2014-15 |
|---|---|---|
| 44 | 44 | 44 |
| Performance Indicators | Targets |
|---|---|
|
The Compliance and Enforcement framework for assisted human reproduction including complaint management is fully implemented and managed. Operational guidelines to administer the Assisted Human Reproduction Act and its associated regulations are well developed and understood and adhered to by the stakeholder community. |
100% of complaints assessed and addressed. Timely and accessible compliance information on the administration of the AHR Act to 100% of the known stakeholder community. Monitoring, assessing and taking corrective action with respect to the prohibitions. Increased collaboration and consultation amongst governments and stakeholders to advance key AHR health and safety issues and facilitate best practices year over year, collectively working to safeguard Canadians who build their families using AHR and those born of the technologies. |
|
Stakeholders are knowledgeable and engaged in AHR issues and activities. Stakeholders are applying evidence-based decision making leading to improved health outcomes. The AHR community shares and exchanges best practices related to principles and science for the benefit all Canadians. |
70% of stakeholders indicate increased knowledge as a result of tools developed, materials published and meetings/forums facilitated by AHRC. Increased number of partnerships and engagement with stakeholders. Increased number of stakeholders using or applying evidence-based tools coordinated by AHRC. Working with the AHR community, decrease the twin rate to 25% in all clinics in Canada by 2012 and 15% by 2015. Increased number of papers, abstracts and presentations on AHR topics. |
|
Support the needs of programs and other corporate obligations of the organization. |
Success of program areas to deliver the Agency's strategic outcome and to achieve their PAA targets. |
| Program Activity | Forecast Spending 2011-12 |
Planned Spending | Alignment to Government of Canada Outcomes | ||
|---|---|---|---|---|---|
| 2012-13 | 2013-14 | 2014-15 | |||
| 1.1 Regulatory Compliance. | 0.8 | 0.8 | 0.8 | 0.8 |
Healthy Canadians |
| 1.2 Knowledge Transfer Program | 1.2 | 1.1 | 1.1 | 1.1 | |
| Total Planned Spending | 2.0 | 1.9 | 1.9 | 1.9 | |
| Program Activity | Forecast Spending 2011-12 |
Planned Spending | ||
|---|---|---|---|---|
| 2012-13 | 2013-14 | 2014-15 | ||
| Internal Services | 1.8 | 1.1 | 1.1 | 1.1 |
| Total Planned Spending | 3.8 | 3.0 | 3.0 | 3.0 |
Note: The Treasury Board authorized spending for the Fiscal Year 2012-13 is $10.5 million, yet the Agency forecasts to spend less than $3 million.
Expenditure Profile
AHRC adheres to prudent fiscal management principles in accordance with the Financial Administration Act, and intends to spend less than its authorized $10.5 million for the 2012-13 fiscal year. With a staff complement of less than 12 FTEs, AHRC plans to spend less than $ 3.0 million to achieve the expected results and strategic outcome "protection and promotion of health and safety of Canadians in relation to assisted human reproduction and related research, within a sound ethical framework."
Departmental Spending Trend
Estimates by Vote
For information on our organizational appropriations, please see the 2012-13 Main Estimates publication.
